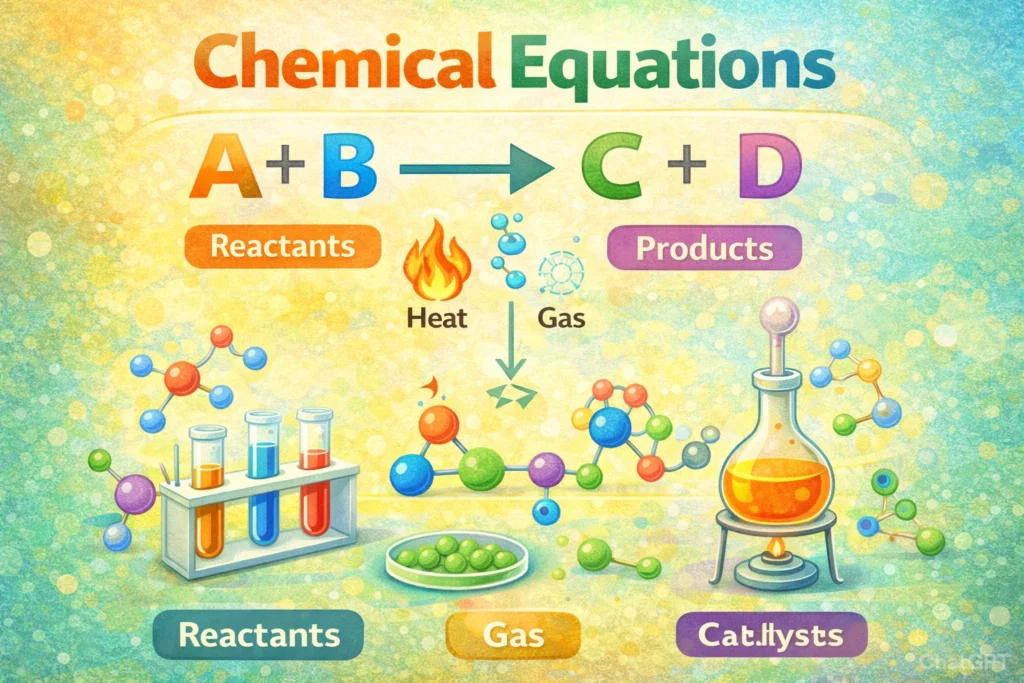
In a chemical equation, the arrow (→) indicates the direction of the chemical reaction, showing that the reactants on the left side transform into products on the right side. It signifies the process of change rather than just a static relationship between substances.
Chemical equations are more than just symbols and formulas. They tell the story of how substances interact, combine, break apart, and transform into entirely new materials. At the heart of every chemical equation is a simple but powerful symbol, the arrow.
The arrow is not just a separator placed between reactants and products. It communicates movement, transformation, and direction. It shows that something is happening. Matter is being rearranged. Bonds are breaking. New bonds are forming. The substances on one side are turning into something different on the other side.
When you see an equation like
2H₂ + O₂ → 2H₂O
the arrow tells you that hydrogen and oxygen react to form water. It points from the reactants on the left to the products on the right. This direction matters. It signals the flow of the reaction, almost like a story unfolding step by step.
In chemistry, the arrow can also carry deeper meaning. A single arrow usually represents a reaction that proceeds primarily in one direction. A double arrow, on the other hand, shows a reversible reaction where products can turn back into reactants under certain conditions. Even small details around the arrow, such as heat, light, or a catalyst written above or below it, provide important information about what is required for the reaction to occur.
In simple terms, the arrow is the bridge between what you start with and what you end up with. It represents change, transformation, and the dynamic nature of chemical processes. Without it, a chemical equation would lose its sense of action and direction.
Why is the Arrow Important?
The arrow in a chemical equation may look simple, but it carries powerful meaning. It does much more than separate substances on the left from substances on the right. It acts as a guide that explains how a reaction progresses, what changes occur, and under what conditions the transformation happens. Without the arrow, a chemical equation would lose clarity and purpose.
Here is why the arrow is so important
1. It Shows the Direction of the Reaction
The arrow indicates the flow of the reaction from reactants to products. Everything written on the left side of the arrow represents the starting substances. Everything on the right side represents the new substances formed after the reaction.
For example
2H₂ + O₂ → 2H₂O
The arrow shows that hydrogen and oxygen move forward in a reaction to form water. It communicates progression and change.
2. It Clarifies What Is Consumed and What Is Produced
Reactants are consumed during the reaction. Products are created. The arrow clearly separates these two roles.
This separation prevents confusion. Without the arrow, it would be unclear which substances are reacting and which are the result of the reaction. The arrow acts like a boundary line with meaning.
3. It Communicates Reaction Conditions
Sometimes symbols or information appear above or below the arrow. These details tell chemists about required conditions such as
Heat
Light
Catalysts
Pressure
For example, a small triangle above the arrow often indicates heat is needed. This makes the arrow a central communication point in the equation.
4. It Represents Reversibility When Needed
In some reactions, the arrow is replaced with a double arrow symbol. This indicates equilibrium, meaning the reaction can move in both directions. Reactants form products, and products can reform reactants.
This small change in arrow style completely changes how the reaction is understood.
5. It Acts as a Roadmap
Think of the arrow as a roadmap for the chemical reaction. It shows where the reaction begins and where it ends. It tells the story of transformation.
Without the arrow, the equation would be incomplete, confusing, and ambiguous. The arrow gives structure, meaning, and direction to the entire chemical process.
Origin of the Arrow in Chemistry
The arrow symbol in chemistry has an interesting history.
- Early 19th Century: Chemists used words like “produces” or “gives” to show reaction outcomes.
- Mid-19th Century: Arrows (→) were introduced as shorthand to simplify complex reactions.
- 20th Century Onwards: The arrow became universal in textbooks, research papers, and lab reports.
The popularity of the arrow comes from its clarity and simplicity, allowing chemists worldwide to communicate reactions efficiently.
Real-World Usage of the Arrow
In laboratory and industrial settings, the arrow is more than a symbol; it guides experiments, synthesis, and manufacturing processes.
Examples of practical usage:
- Pharmaceutical industry: Tracking drug synthesis reactions.
- Environmental chemistry: Representing pollutant breakdown.
- Education: Teaching students reaction pathways.
Using the arrow correctly ensures safety and accuracy in chemical work. Misplacing or misinterpreting it could lead to errors in calculations, reactions, or reporting.
Examples of the Arrow in Chemical Equations
Here are a few common types of reactions and how the arrow is used:
| Reaction Type | Equation Example | Explanation |
|---|---|---|
| Synthesis | N₂ + 3H₂ → 2NH₃ | Nitrogen and hydrogen combine to form ammonia. |
| Decomposition | 2H₂O → 2H₂ + O₂ | Water breaks down into hydrogen and oxygen gases. |
| Single Replacement | Zn + 2HCl → ZnCl₂ + H₂ | Zinc displaces hydrogen from hydrochloric acid. |
| Double Replacement | AgNO₃ + NaCl → AgCl + NaNO₃ | Ions swap partners, forming new products. |
Friendly Example: When baking soda reacts with vinegar:NaHCO3+CH3COOH→CO2+H2O+NaCH3COO
The arrow shows the transformation from ingredients (reactants) to fizzing gas and other products.
Neutral Example: Iron rusting over time:4Fe+3O2→2Fe2O3
Here, the arrow simply indicates the chemical change without emotion.
Negative/Dismissive Example: Some students see the arrow as just “a line,” ignoring its role in direction and process, which can lead to mistakes in balancing equations. ⚠️
Comparison with Related Symbols
The arrow is not the only symbol in chemical equations. Here’s how it compares:
| Symbol | Meaning | Usage |
|---|---|---|
| → | Produces / yields | Most common for reactions that go forward. |
| ⇌ | Equilibrium | Reversible reactions, e.g., N₂O₄ ⇌ 2NO₂ |
| ↑ | Gas evolution | Indicates a gas is released, e.g., H₂↑ |
| ↓ | Precipitate formation | Indicates a solid product forms, e.g., AgCl↓ |
| →(Δ) | Heat applied | Reaction requires heating, e.g., CaCO₃ →(Δ) CaO + CO₂ |
Tip: Understanding these variations helps interpret reactions more accurately, especially in labs and exams.
Alternate Meanings of the Arrow
While in chemistry the arrow usually means “produces,” it can have context-specific meanings:
- Equilibrium conditions: ⇌ shows reversible reactions.
- Reaction conditions: Arrows with symbols above or below indicate catalysts, light, or temperature, e.g., →(Pt) shows platinum is used.
- Energy diagrams: Arrows may show activation energy or direction of electron flow.
Knowing these alternatives prevents confusion when reading advanced chemistry materials.
Professional and Polite Alternatives
In written communication, you can replace the arrow with words for clarity, especially in educational or public-facing materials:
- “yields” → e.g., 2H₂ + O₂ yields 2H₂O
- “produces” → e.g., Zn + HCl produces ZnCl₂ + H₂
- “forms” → e.g., C + O₂ forms CO₂
These alternatives are particularly useful in textbooks, presentations, or when explaining chemistry to beginners.
Common Mistakes to Avoid
- Ignoring the arrow’s direction – Reactants must be on the left, products on the right.
- Confusing reversible reactions – Use ⇌ for equilibrium instead of →.
- Misplacing reaction conditions – Heat, catalysts, or light should be clearly indicated.
- Overlooking gas or precipitate indicators – ↑ or ↓ is essential in some reactions.
Remember, the arrow is more than a symbol; it communicates the chemistry process itself.
Example Table – Arrow Usage in Different Contexts
| Reaction | Arrow Meaning | Notes |
|---|---|---|
| H₂ + Cl₂ → 2HCl | Produces | Simple one-way reaction |
| N₂ + 3H₂ ⇌ 2NH₃ | Reversible | Ammonia synthesis under equilibrium |
| CaCO₃ →(Δ) CaO + CO₂ | Produces with heat | Thermal decomposition |
| Zn + HCl → ZnCl₂ + H₂↑ | Produces gas | Gas evolution indicated |
Practical Tips for Students and Chemists
- Always place reactants on the left and products on the right.
- Pay attention to additional symbols around the arrow.
- Use the arrow consistently in lab reports to avoid confusion.
- When teaching or explaining, consider writing “yields” instead of → for clarity.
FAQs
1. What does the arrow (→) represent in a chemical equation?
It represents the direction of the chemical reaction, showing that reactants transform into products.
2. Can a chemical reaction occur without an arrow?
No, the arrow is essential to indicate which substances are changing and in which direction the reaction proceeds.
3. What is the difference between → and ⇌?
→ indicates a one-way reaction, while ⇌ indicates a reversible reaction at equilibrium.
4. Why are there sometimes symbols above or below the arrow?
They indicate reaction conditions, like heat (Δ), a catalyst (Pt), or light (hv).
5. Does the arrow mean the reaction is instantaneous?
Not necessarily. The arrow only shows direction; the speed of the reaction depends on conditions and kinetics.
6. Can the arrow point both ways?
Yes, in reversible reactions, represented by ⇌, indicating products can revert to reactants.
7. How do you read the arrow in words?
It can be read as “produces,” “yields,” or “forms” depending on the context.
8. Are there alternative symbols for the arrow?
Yes, depending on the reaction type, you might see ↑ for gases, ↓ for precipitates, or → with conditions like heat or catalysts.
Conclusion
The arrow in a chemical equation is a simple yet powerful tool that communicates change, direction, and reaction outcome. From basic school experiments to industrial chemical synthesis, understanding the arrow ensures clarity and accuracy.
Always remember:
- It shows direction from reactants to products.
- Variants like ⇌, ↑, ↓ provide extra information about reversibility and physical states.
- Alternatives like “yields” or “produces” are useful for communication in education or public writing.
Mastering the arrow is a small step that leads to big clarity in chemistry. ✅
Read More Related Articles:
- “Le” Means in Spanish: Why This Small Word Matters So Much In 2026
- OP Meaning in Gaming – The Term Every Gamer Uses (2026)
- HU Meaning in Text: Simple Guide with Examples You Can’t Miss (2026)

Neon Samuel is a digital content creator at TextSprout.com, dedicated to decoding modern words, slang, and expressions. His writing helps readers quickly grasp meanings and understand how terms are used in real conversations across text and social platforms.